Let me start by saying I truly love the Daily Show. Jon Stewart cracks me up, and together with MSN (my homepage) and Facebook, he’s one of my three main sources of political news. His focus, of course, is politics not science, and that’s why although his satire is reliably hilarious, when it comes to scientific & public health controversies he tends to shoot a little wide of the mark.
Take, for example, the chemical azodicarbonamide. Recently Stewart decried its presence in Subway sandwich bread because it’s also found in yoga mats, which to him proved it was dangerous — no more needed to be said. But wait a minute. Gypsum is both a food additive and an industrial chemical used to make drywall; does Stewart think it should be banned too? While we’re at it why not ban salt? they use that to make chlorine gas through electrolysis, dontcha know. Now there’s some scary stuff.
As it happens, I agree removing azodicarbonamide from bread is probably a good idea because it’s not really necessary and if a food additive is both questionable and not really necessary, why not take it out? makes life simpler. On the other hand, however, although I think taking it out is a no-brainer, at the same time I have to point out the evidence to show azodicarbonamide is dangerous at 45 parts per million (the FDA-mandated limit) isn’t exactly overwhelming. So why did Subway decide to remove it now, and how did Jon Stewart hear about it? It turns out their decision was prompted by a petition circulated by a food blogger I’d never heard of called The Food Babe (and yes, Food Babe is what she calls herself).
The wonderful thing about the Internet is that it makes it so easy to access information — and of course that’s the downside to the Internet too. It only takes a click and a few spare minutes to satisfy your curiosity, with the result that you end up learning about all kinds of things you didn’t really need to know, like Katie Perry’s love life and which celebrities have cool tattoos. This is especially true if you’re at work surfing the internet on your lunch. That was how I ended up reading through a few posts from The Food Babe’s blog, which claims four million followers all told. In retrospect I wish I’d kept reading about Katie Perry’s love life instead.
Food Babe is on a mission to find out what’s in your food. That’s commendable in itself and some of what she’s doing is interesting. The problem is she blends information with misinformation all over the place. Take, for example, this article about What’s In Your Tea. Food Babe points out that some tea bags are made of plastic like nylon or polylactic acid (a corn-based biodegradable plastic) while others are made from paper, and the paper-based bags are treated with polyamide-epichlorohydrin resins. So what the hell is that? and why would anybody put it in a teabag? Well, let me explain.
The 3D structure of epichlorhydrin is like this:
 Think back to my last post about the West Virginia chemical spill. Remember that a carbon atom with four bonds wants to have its four bonds arranged so it’s at the center of a tetrahedron. You can rationalize this by remembering that bonds are pairs of shared electrons and electrons repel each other so bonds want to be as far away from each other as they can*.
Think back to my last post about the West Virginia chemical spill. Remember that a carbon atom with four bonds wants to have its four bonds arranged so it’s at the center of a tetrahedron. You can rationalize this by remembering that bonds are pairs of shared electrons and electrons repel each other so bonds want to be as far away from each other as they can*.
Look at the two carbons(black) in that three-membered ring or triangle up at the top. Because they’re part of a triangular three-membered ring their bonds to the oxygen atom(red) are forced into a shape they’d rather not assume and are strained. The strain makes those bonds easier to break in the same way bending a piece of plastic out of shape makes it easier to snap.
That might not matter so much if it weren’t for the oxygen atom. Remember that as you go towards the upper right-hand corner of the periodic table elements get more selfish about how they share electrons. Oxygen is much more selfish than carbon so the two carbons in this ring are electron-poor while the oxygen is electron-rich. The combination of uneven electron density together with the strained bonds means this two-carbon one-oxygen triangle is very reactive. This type of ring is called an epoxide.
Almost any time you see an epoxide you can bet the carbon atoms in it want to react with things that are electron-rich, like nitrogen atoms in DNA. The exception is if the epoxide ring has lots of bulky carbon chains on it that block electron-rich stuff from reacting with it. With an unencumbered epoxide ring like this one, however, it’s pretty reactive. That’s why many epoxides are highly toxic and/or carcinogenic. Take this one for example:
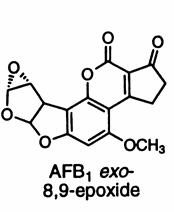 There are some molds that grow on peanuts and produce chemicals called aflatoxins. When your liver gets ahold of aflatoxin it modifies it to make the compound you see above. Notice how it contains an epoxide ring? That epoxide ring can react with DNA, and that’s why aflatoxins are carcinogens. They’re entirely natural carcinogens, by the way; they’re made by molds found in nature, and peanut butter may contain aflatoxins at trace concentrations because it’s damn near impossible to remove them completely, although the amount peanut butter can contain is strictly regulated by FDA. Once again, “Mother Nature” turns out to be less friendly than she appears. Why this crazy mold wants to give us cancer nobody knows. But anyhow. Let’s get back to epichlorohydrin.
There are some molds that grow on peanuts and produce chemicals called aflatoxins. When your liver gets ahold of aflatoxin it modifies it to make the compound you see above. Notice how it contains an epoxide ring? That epoxide ring can react with DNA, and that’s why aflatoxins are carcinogens. They’re entirely natural carcinogens, by the way; they’re made by molds found in nature, and peanut butter may contain aflatoxins at trace concentrations because it’s damn near impossible to remove them completely, although the amount peanut butter can contain is strictly regulated by FDA. Once again, “Mother Nature” turns out to be less friendly than she appears. Why this crazy mold wants to give us cancer nobody knows. But anyhow. Let’s get back to epichlorohydrin.
Notice the green chlorine atom down at the corner. Chlorine is much more electron-hungry than carbon. It’s also much bigger than carbon because it’s a whole row further down in the periodic table. When you have a carbon that has all single bonds attached to a chlorine, the bond between them is long and weak, and since the chlorine is hogging the electrons they share, that carbon is tempted to react with anything really electron-rich, kicking the chlorine atom out of the molecule in the process.
Based on its structure, you would predict that epichlorohydrin should be highly toxic and carcinogenic because the unencumbered epoxide ring and the chlorine-on-a-single-bonded-carbon are pretty reactive in ways we don’t like. And you’d be right. Lab tests have shown it’s exactly that.
So what kind of moron would put epichlorohydrin anywhere near a tea bag? Well, here’s where the story gets more complicated. Make a tea bag out of a plastic like nylon and it’ll hold together ok in water. Paper on the other hand won’t. If you put paper in water it gets all soggy and falls apart. To make paper water-stable you have to treat it with something. Many manufacturers use a polymer called a polyamide-epichlorohydrin resin, which is made by taking epichlorohydrin and reacting it with a polymer that contains multiple amine or nitrogen groups. The amine groups are electron-rich, so they react with epichlorohydrin molecules and thus become part of a long chain with reactive side chains stuck onto it. Treat paper with the resulting polymer and you make it more water-stable**.
If the epichlorohydrin is used up in the reaction, then who cares? Trouble is any remaining epichlorohydrin can react with water to form this critter:

The epoxide ring in epichlorohydrin is so reactive it reacts with water molecules. This end product is called 3-MCPD and since it still contains a chlorine atom connected to an all-single-bonds carbon, it can react with electron-rich stuff; your body metabolizes it in part by knocking off the chlorine and converting some of it back to an epoxide called glycidol which (like most epoxides) is bad news. Based on the structure you’d predict 3-MCPD is carcinogenic, and the State of California says based on available evidence they’re pretty sure it is carcinogenic like you’d expect. So I assume Food Babe doesn’t like epichlorohydrin in tea bag production because you get trace levels of 3-MCPD at the other end, and that residual 3-MCPD probably ends up in your tea. That’s a fair concern and definitely worthy of investigation.
Food Babe didn’t go into any of this, just basically said tea bag manufacturers use epichloroydrin which is dangerous so that’s bad, but hey, same difference. It’s cool she brought this up because it’s something you probably don’t think about a lot unless you’re into chemistry or something. (How come a paper tea bag holds together when paper usually falls apart in water, hmmmm?) She also raises some legitimate concerns about whether pesticide residues are found on certain tea brands and if so at what levels. Which is also stuff worth thinking about.
Now I don’t drink a lot of tea. But if I did and I were looking to avoid this 3-MCPD issue, I would say that teabags made from polylactic acid would be a better way to go. I’m not a big fan of nylon because it ends up in landfills and isn’t very biodegradable; polylactic acid is made from corn and is much easier for microbes to break down, so as an environmentalist that makes me happy. Food Babe, however, doesn’t like polylactic acid because it’s made from genetically modified corn, which is (she says) unnatural. That’s where she and I part company. If you’ve read any of my past posts about GMOs and “natural=good” you’ll know why. And her article just goes downhill from there.
Food Babe quotes Dr. Mercola, IMHO one of the quackiest rogue doctors on the web, about the safety of nylon in teabags. For starters, if you’re using Dr. Mercola as a source, you need a factchecker in the house. Mercola is a dude who makes money by arguing that vaccines and traditional medicine are harmful and that his “natural supplements” are a better way to go. The FDA had to send the guy warning letters because his website claimed his infrared camera was a “safe cancer screening tool” that could “detect hidden inflammation“. This doctor-turned-supplement-guru is such a joke he has his very own page on the quack-tracking website Quackwatch. And leaving aside the source, her argument as to why nylon is bad is ill-informed:
“While the plastic itself won’t melt in your tea, the glass transition temperature could potentially leak out harmful phthalates if there are such things in your tea.”
You don’t use phthalates to make nylon. Maybe Food Babe should find out what nylon actually is before she starts telling us why it’s such a terrible thing. Actually nylon is reasonably harmless stuff, Food Babe to the contrary. My only beef with it would be that like most plastics it’s not terribly biodegradable. Given that most teabags probably end up in landfills that’s kind of a problem.
And then there’s this, which is so hopeless it made me want to stop reading:
“Furthermore, a majority of Teavana teas contain added flavor – specifically “artificial flavoring”. If their tea is so high-end, why would they be adding ingredients produced by fractional distillation and chemical manipulation of various chemicals like crude oil or coal tar? Coal tar in my tea? No, thanks.”
Crude oil and coal tar are sources of hydrocarbons you use as building blocks. In the course of those reactions those building blocks are converted into new molecules with entirely different properties from the original building blocks. Assuming that something is bad because it was made from hydrocarbons that came from crude oil is ridiculous. You might as well argue the water you drink is bad because it was dinosaur urine once upon a time. Well, yeah, sure, but it’s come a long way since then, you know.
The remarkable things that Food Babe believes are so numerous I don’t have time to go through most of them. Here’s a few examples that really bothered me. Food Babe believes that propylene glycol is toxic because it’s found in antifreeze. What she really means is ethylene glycol, which has two carbons compared to propylene glycol’s three — and believe it or not that makes a huge difference. Food Babe is also not a fan of potassium sorbate because it’s a preservative. Now there are preservatives I don’t like, but I have nothing against potassium sorbate. Here’s the structure:
 Any time you see that COO- group with a straight chain of hydrocarbons attached to it, you’re looking at a fatty acid. If that fatty acid contains double bonds it’s unsaturated, while if it’s all single bonds it’s saturated. So potassium sorbate is an unsaturated fatty acid. Your body handles it the same way it would any other fat: fuel. It’s fat. It’s something you can burn. So potassium sorbate is relatively harmless from your point of view; your body will just take it and burn it for fuel. It makes mold grow more slowly and that’s why we use it as a preservative. Why Food Babe doesn’t like it I have no idea.
Any time you see that COO- group with a straight chain of hydrocarbons attached to it, you’re looking at a fatty acid. If that fatty acid contains double bonds it’s unsaturated, while if it’s all single bonds it’s saturated. So potassium sorbate is an unsaturated fatty acid. Your body handles it the same way it would any other fat: fuel. It’s fat. It’s something you can burn. So potassium sorbate is relatively harmless from your point of view; your body will just take it and burn it for fuel. It makes mold grow more slowly and that’s why we use it as a preservative. Why Food Babe doesn’t like it I have no idea.
And this is far from the worst of it, because as it turns out Food Babe doesn’t like the flu shot, either. Now there are things not to like about the flu shot. The flu shot only protects against a few strains, the ones CDC thinks are most likely to be running around in a given year, and sometimes they get that wrong because the virus is so freaking unpredictable. You could argue that if you’re a young healthy adult like Food Babe, you’re not worried about dying from flu, so you’re not going to take a shot that may or may not protect you from a disease that is unlikely to kill you anyway. And that would be a fair argument to make. Before you doctors out there start giving me a hard time, however, please bear in mind, I’m not saying I agree or disagree, I’m saying that’s a fair argument we can debate. But that’s not why Food Babe doesn’t like the flu shot. Food Babe doesn’t like it because it contains toxic chemicals like formaldehyde and aluminum salts.
Never mind that Food Babe’s blood naturally contains many times more formaldehyde than you’ll find in a vaccine, and never mind she’s exposed to aluminum in nearly everything she eats because aluminum is found in dirt and many plants are reasonably good at taking up small amounts of it. Food Babe doesn’t know or doesn’t care. She just knows the flu shot isn’t natural. Presumably Food Babe uses cars, condoms and computers just like me, and presumably she knows those aren’t natural too. But somehow the irony is lost on her.
So this is why I should have kept reading about Katie Perry’s fascinating love life instead of googling Subway and azodicarbonamide, which led me by a few short clicks to Food Babe’s blog. Food Babe’s blog is frustrating to read because she’s writing about a subject with so much potential and yet so much of it is wrong. It really would be fascinating to discuss in detail all the stuff that’s in your food. There’s a lot of things in there you don’t know or think about. Take this business with the epichlorohydrin-polyamide resins; or the natural carcinogens produced by molds on moldy peanuts. Those are things you probably didn’t know, right? It would be very cool for someone to go out there and blog about all this kind of stuff; take a close look at what’s really in your food and how it’s made. It would be a real eye-opener. Unfortunately, that’s not what Food Babe is doing.
She’s just feeding her four million followers more of the same old Mother Nature Knows Best crap — if it’s not natural, don’t buy it. And that’s why her blog is such a seamless blend of interesting facts and mindless ignorance. That’s why she can simultaneously slam polyamide-epichlorohydrin in tea bag manufacture (which might be something worth criticizing) and the flu vaccine (which has been extensively tested for safety and is certainly not).
There’s something I was told when I was a kid that I’ve always found true: life is complicated, and the closer you look at it the messier it gets. There are no easy answers. This is true of science just as it is of politics or relationships or really anything else. If you meet someone who claims they have a simple answer that solves everything, they’re probably trying to sell you something. Unfortunately we humans are attracted to easy answers and simple solutions, which is how politicians and self-help gurus and snake oil salesmen make a living. Your life would be great if you’d learn these simple laws of success. Elect me as president and the Change will begin. Eat all-natural foods and you’ll be healthy forever. It sounds nice. If only it were true…
———————————–
*This is how the valence-shell electron pair repulsion or VSEPR model explains the tetrahedral shape. VSEPR is nice because it’s simple and enables you to quickly figure out a molecule’s shape in your head. For the chemistry undergrads out there, however, remember that VSEPR is just a nice little qualitative predictive tool and nothing more…quantum chemistry provides a more accurate explanation for why a molecule has the shape it does. The closer you look the more complicated it gets as usual.
**There is still some debate over what’s going on here — are the side chains in the polymer reacting with hydroxyls on cellulose or are the polymers becoming cross-linked and wrapping around cellulose fibers to hold them together? Either way, it works.

Thank you very much for the awesome article!
While your article is insightful I think you are missing the point.
By your own point life is very complex, and sometimes (particularly over long period of times) science gets it wrong. When complex systems are involved mistakes are made. Any scientist that is being honest with himself/herself realizes that this can occur.
In addition science can occasionally be bought. Studies influenced in such a way cannot be trusted, and although it may not be common it does happen.
All of that being said the push for natural food isn’t about keeping one healthy forever. It’s about minimizing the risk from science being wrong. The theory is if you are eating food that several hundred generations of our ancestors ate for thousands of years, minus the less natural additives that many of those risks fall to the wayside.
Excellent point however a large number of our ancestors died very young by our standards. Also as technology and understanding advance we gain better insight into things like food. I certainty agree and also stay away from chemicals as much as possible but there is a point where you go from being healthy to being unhealthily obsessed. An excellent debate for sure and one which I will now have to bow out of as I don’t have enough insight to comment beyond asking all involved to keep an open mind.
Sure many of them died very young, and modern meds have done much to increase our lifespan. Obviously when presented with “take this experimental drug and you might still live” most people when given the choice will take the drug.
In the case of the food additives though its very different.. You really don’t need most of them in food. Beef is beef without dye being injected into it. Bread can consist of as little as flour, water, and yeast.. Yes it won’t be shelf stable, but its a relatively simple list.
I appreciate the response to the comment. Lets see how long we can keep this civil 😀
Perfectly said. I couldn’t agree more. If the additives (which are not needed) aren’t doing any good, they are probably doing bad, rarely does something do nothing.
I agree and I disagree — for two reasons.
First, the argument that we humans evolved to thrive on a certain diet could be true although I don’t know that for sure; frankly it looks like humans evolved to exhibit remarkable flexibility in terms of what we can eat. Even if it is true, however, it’s not clear that diet is anything we can reproduce in the modern world. Nearly all the fruits, vegetables and animals you eat have been subjected to many generations of intensive breeding to give them the shape/consistency you now see. The wild ancestors of tomatoes, bananas and corn look almost nothing like the corn, tomatoes and bananas you eat today. It might be nice in theory if we could all go back to some hunter-gatherer diet of raw berries, nuts and whatever you can hunt/trap, but it’s totally impractical. We are where we are and in most ways that’s a good thing — the average hunter-gatherer life expectancy wasn’t all that long.
You point out that humans are probably better equipped by evolution to deal with the chemicals we regularly encounter in Nature than we are with artificial chemicals that aren’t found in nature. Probably true — but still not a good reason to assume all artificial chemicals are bad or all natural chemicals are good. Each chemical has its own individual properties determined by its structure, and there are plenty of things in Nature that want to poison us, like the molds that make aflatoxins or the bacteria that cause botulism.
But I DO agree wholeheartedly with the point you’re making about unnecessary food additives. A lot of the additives you have in food aren’t really totally necessary if you get down to it, and if they’re not totally necessary why not take them out just on general principle? Take food coloring for example. Now the food colorings currently on the market have been extensively studied by FDA because of a scare back in the day over a now-banned orange dye and I’m reasonably convinced they’re safe. But are they necessary? No. You don’t NEED your food dyed all kinds of pretty colors. So why not take them out? This is the same for azodicarbonamide. I’m fairly confident for myself based on what I know about its chemistry that FDA is prolly correct when they say it’s safe below 45 parts per million; but you don’t really need to have it in bread. So why not take it out? If something doesn’t need to be in there, then pull it. Makes life simpler. The problem, of course, is that additives like dyes or azodicarbonamide give food attributes that make it more appealing to consumers — they sell, in other words — and companies are understandably reluctant to do something that would make their product less marketable.
I think y’all need to google botulism and trichinosis, to say the least. There’s a reason why meds, antibiotics, advanced preservation processes and “science” in general works. Edumacate yourselves people, plenty of history to prove you naysayers wrong.
Very interesting and well researched article. Very important topic as well. Much appreciated
If you don’t want gypsum in your bread, and you agree with his remarks on azodicarbonimide, then the balance of your article is baloney.
My article isn’t really about Stewart per se (maybe I should have been more careful about the title). It’s about the arguments that he and Food Babe used and why they’re flawed. Food Babe and Stewart and many others are starting from the assumption that if a chemical or ingredient has a long name, is made in a lab, is used in an industrial process or is not found in nature, it must be dangerous. As Food Babe’s blog illustrates, if you follow this logic to its conclusion you would avoid GMO technology (which IMHO has incredible potential to improve agriculture), vaccines (which are very safe and have saved countless lives) and modern medicine (which has again saved many lives and is a lot better than the traditional alternative) — and Food Babe does indeed eschew or at best mistrust all of the above. That’s why I feel it’s important to refute Food Babe & Stewart’s starting assumption, because this whole natural=good artificial=bad dichotomy that’s so ingrained among environmentalists fuels anti-vaccination paranoia and retards progress.
Loved the article and enjoy your comment answers.
But I’d like to quibble with your lack of Oxford commas. 😉
So Puff….here’s the deal…I’m a woman of average intelligence, but VERY interested in researching what I’m eating. YOU lost ME at the first diagram. Perhaps if you could simplify the whole deal, I’d understand better. As far as clarifying better what your article was TRULY about – why do that when you can sucker just about everybody into at least clicking on it and do a short skim of the article…shame on you. If you can’t bet readers because of the subject, then re-work the content. I admit it…you got me because of Food Babe. BUT – she got me to read HER blog because she didn’t hide that it is about the chemicals in our food. She got me interested in researching about it. Yep…she may not be correct 100% of the time, but at least she’s honestly and simply putting out there for people to decide for themselves. IF I could comprehend the information you put forth, I might research that also. But, for right now, it’s nothing but a bunch of $5 words strung together to resemble a sentence, with diagrams included.
Sorry, I’ll stick with Food Babe.
Precisely!
My “precisely” comment above was in response to Joe’s comment. It really is not so very complex however tons of money is made by marketing and promoting all of it as such. Oh and btw, Quackwatch is a sham; even one without a biotech degree can discern truth vs. propaganda. You can keep your GMO’s, flouridated water, toxic food additives and more, thanks.
And Audrey, you are very wise indeed.
The article contains some insightful explanations of why certain chemicals are bad for us.
However, the following analogy really decrease the credibility of the whole article and to certain extent the intelligence of the author:
“She just knows the flu shot isn’t natural. Presumably Food Babe uses cars, condoms and computers just like me, and presumably she knows those aren’t natural too. But somehow the irony is lost on her.”
Why? Because the author used real world examples that applies to context of real life and what real people use and partake in throughout their lives? Your comment reduces the credibility of YOUR intelligence becase such a comment lacks in foresight and fails to provide any sort of objectivity or even a qualitative basis of reasoning.
i highly doubt these people care what the actual scientific truth to these issues is. they are in the business of generating page view and maybe selling a few books. their argument against azodicarbonamide or whatever the cause of the day is doesn’t have to be airtight, it just has to be substantial enough to not be discredited instantly and simply. if it takes you longer to explain why she’s wrong than it took her to say “this stuff is in yoga mats, that’s gross!” you’ve already lost the attention of her readership and they are either closing your tab or scrolling past the rest of your expostulation to tell you why you’re wrong
the food babe and her ilk don’t mind being wrong on the science at the final evaluation, as long as they were the focal point for attention paid the issue. at this point it’s her job to come up with these viral campaigns about food ingredients, and she’s likely vetting new prospective issues continuously. if she doesn’t come up with something that’s actually important, it will just be the next best thing, i.e. some shoddy paper linking mega-doses of aspartame to cancer or what-have-you
personally i’m fine with these kinds of movements bring about the banning of a lot of dyes and additives from food, even if for the wrong reason. there’s entirely too much deception in food marketing
That’s a good point. Given how much misinformation there is in food marketing (and yeah, agree completely there’s a lot), if Food Babe and her ilk go around causing unnecessary panic for all the wrong reasons, it may actually be a good thing if it forces these companies to be more careful about how they market their wares.
But wouldn’t it be great if they could facilitate or drive positive change using correct information though!
The point is not that she “blends information with misinformation”. She’s an activist not a reporter.
Reality is “the scientists, aka FDA and CO” have failed the American public. Congrats to them for “being right” and not get anything done about it. Ingredients long time forbidden elsewhere are the norm here in US. Organic food 2-3 times more expensive than “regular food” which is more expensive than “junk food”. Children obesity rates. The list goes on ….
So I’m asking the readers, what’s more important, being always right and not do s..t about it or acting sometimes based on false premises ?
If you are an activist it is even more important that you get your facts right, because at least part of your mission is to educate. Education seems to me to be the dominant theme with Vani Hari. To answer your question to the readers for myself: acting on false premises in this way through ignorance is negligent, and acting knowingly is deception. If you are an activist and you care then the truth is your sword, and incorrect information and hyperbole blunt it.
Good article, but I don’t think someone posting on a sight called “the Puff The Mutant Dragon” has much room to critic someone going by the name “The Food Babe”. Just sayin’….
I dont think someone posting a comment with either themselves wearing a red clown nose, or a picture of someone else wearing it has much room to critize the choice of a blogger either. Just saying
“…I agree removing azodicarbonamide from bread is probably a good idea because it’s not really necessary and if a food additive is both questionable and not really necessary, why not take it out?”
Experimental research has shown time and again that bread made from flour with azodicarbonamide is more visually appealing and has a more pleasant texture, while being no less healthy than its azodicarbonamide-less counterpart. “Not necessary?” Well, yes, but we all enjoy lots of things that aren’t really necessary. Is chocolate necessary? And,collectively, as consumers, Americans have said that they prefer bread with this particular, harmless additive. Gawd, I wish we had bread with azodicarbonamide down here in Australia.
I would be a bit wary of PLA teabags, but then again I’ve been using PLA and other plastics in 3D printing for years, so I understand their proerties quite a bit (compared to the average lay-person).
PLA melts at a very low temp naturally, so you have to wonder what they’ve done to it to make it withstand the temp of boiling water. This usually ends up being all sorts of fillers that help raise the temp, usually by keeping all the polymers wrapped around each other when they want to slide apart. Usually these fillers are not very nice stuff.
But as for nylon, as long as it’s cleaned properly and not exposed to seriously high temps (200C+), I believe the chances of any degradation are so minor it’s pretty much undetectable.
PS: Great article. Keep up the good work.
They actually do use propylene glycol for antifreeze… when they want to make a non-toxic antifreeze! (For use in winterizing drinking water lines, for instance.)
Reblogged this on Thoughts, Theories, Facts, and Notions and commented:
At heart, I am a scientist. This blog makes me proud. Using the known universe to learn about the rest of the known universe. A plodding methodical set of steps in the best direction possible until more information provides a better direction.
Great post, and I understand your frustration. I teach organic chem and currently have a student who insists on preaching the gospel of natural, and just takes it tooo far sometimes. Her last outburst was a diatribe on the evils of vanillin…because it was “synthetic”. A fellow student lost patience and filled her in on what the compound is and that it is also produced naturally. Gasp!! A revelation!
You keep up the good work, Puff.
Great post and some excellent, simple explanations of the related chemistry: it all helps to counter the illogical “all chemicals are bad” message!
The best solution, of course, is to use no teabag at all. Surely a connoisseur would use only loose tea!
Would love your input on the ebola outbreak.
Woman is a fear monger who hawks products. She is a fraud and the only reason people pay attention to her because she is attractive
Why don’t you make a Food Blog with info on all the latest controversial chemicals?
I really enjoyed reading this. As someone who follows “Food Babe” on Facebook, and has read some of her blogs, I know that what she has written always needs to be taken with a grain of salt. I don’t agree with a lot of her statements and it’s painfully obvious that she’s promoting certain products for a definite payout… But if you look closely, there’s some interesting info between the lines… Like most sites, she’s got an agenda and she’s doing her damnedest to promote it.
I agree wholeheartedly that it would be excellent if someone could write about the nature of our food from an unbiased viewpoint, but let’s be honest… Will that ever happen? Which is why, when people use the web for research, they usually end up with ten different web pages open on the same topic… And each one claims something different. We all just try and do the best we can with what we have… Which is why I enjoyed reading your post so much. It was very informative and lacked the usual one-sided feel. Also missing was the normal anger that accompanies both sides of the gmo/non-gmo debate. If only everyone conversed this way, I’m certain there would be much less animosity.
Please write a new article soon!!
Excellent post but now I am all pissed because I have been drinking tea in tea bags for YEARS all the while thinking I was drinking something good fro me…frack!
Yoga Health Solution
http://www.yogahealthsolution.com/
Fitness trainer at home in delhi
http://www.yogahealthsolution.com/
How Yoga Transforms Lives. The Gym Transformed My Body, But Yoga Changed My LIFE
#Yoga #yogahealthsolution
http://bit.ly/2drXDTD
http://www.yogahealthsolution.com/
I have noticed you don’t monetize your website, don’t waste your traffic, you can earn additional cash every month.
You can use the best adsense alternative for any type of
website (they approve all websites), for more details simply search
in gooogle: boorfe’s tips monetize your website